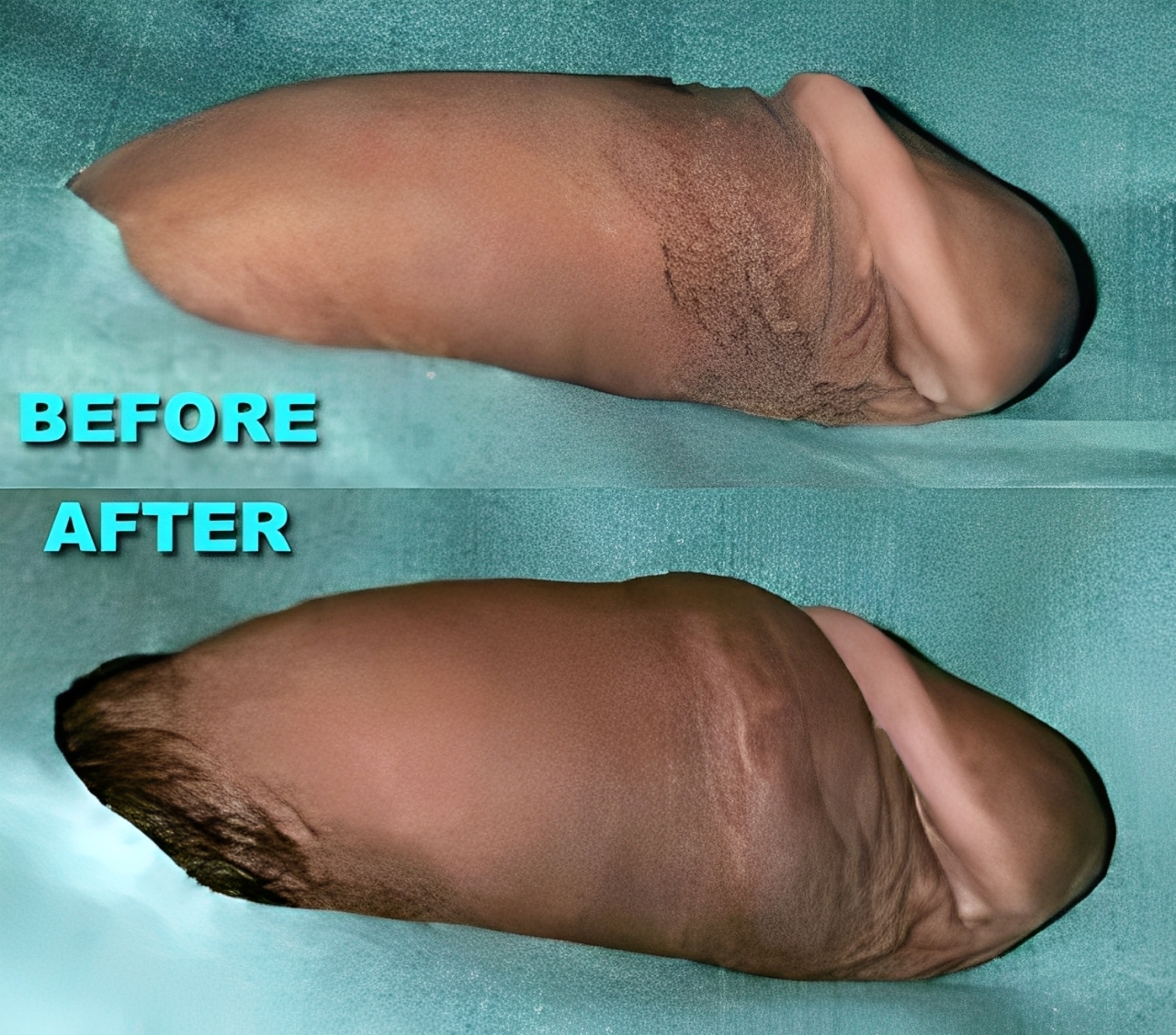
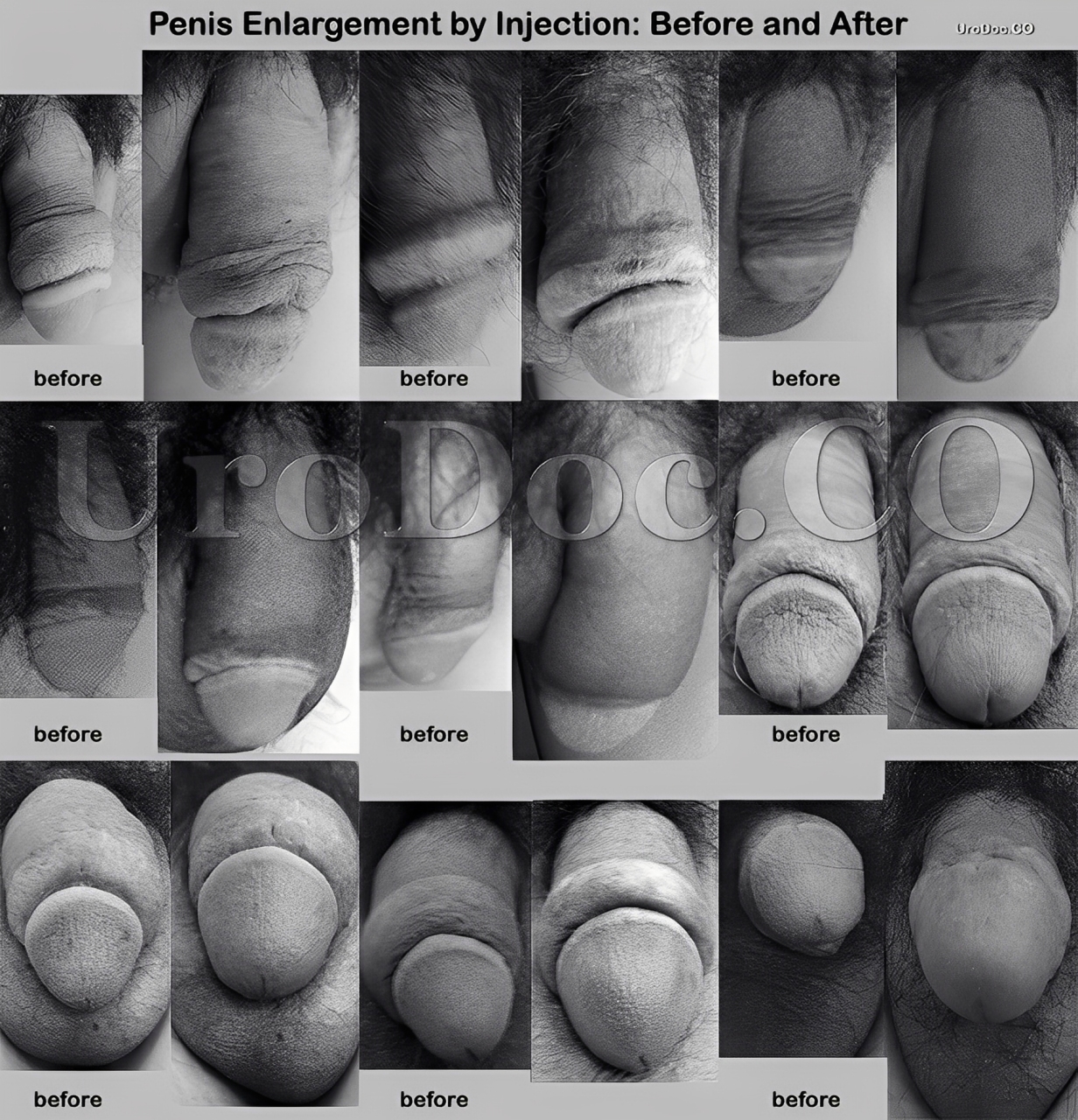
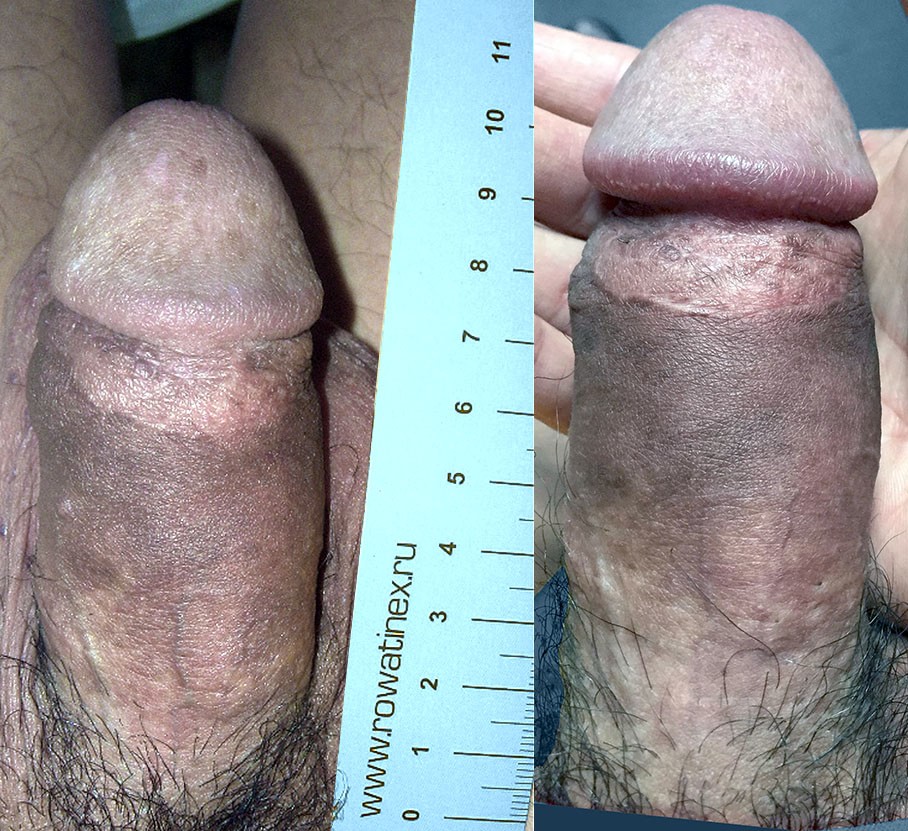
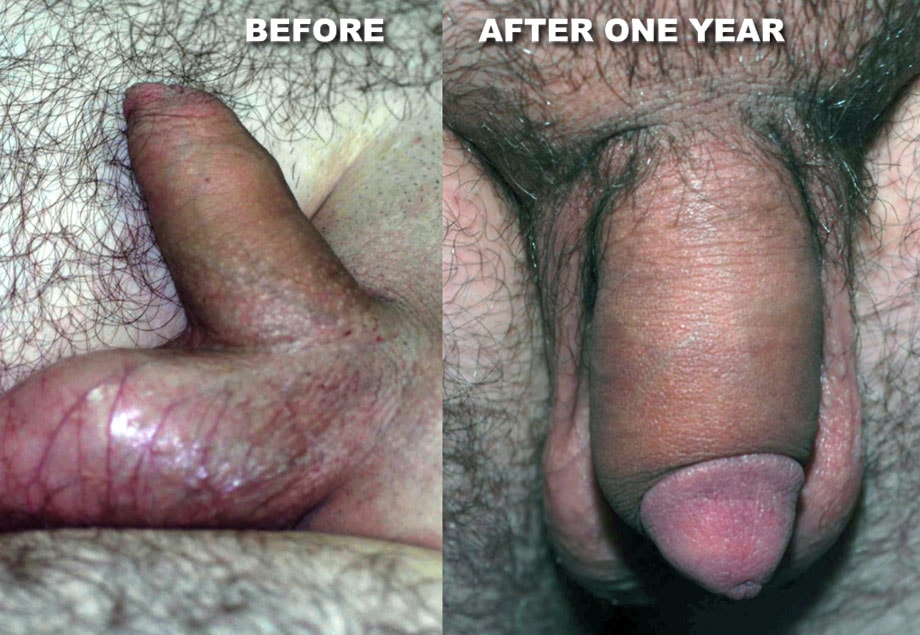
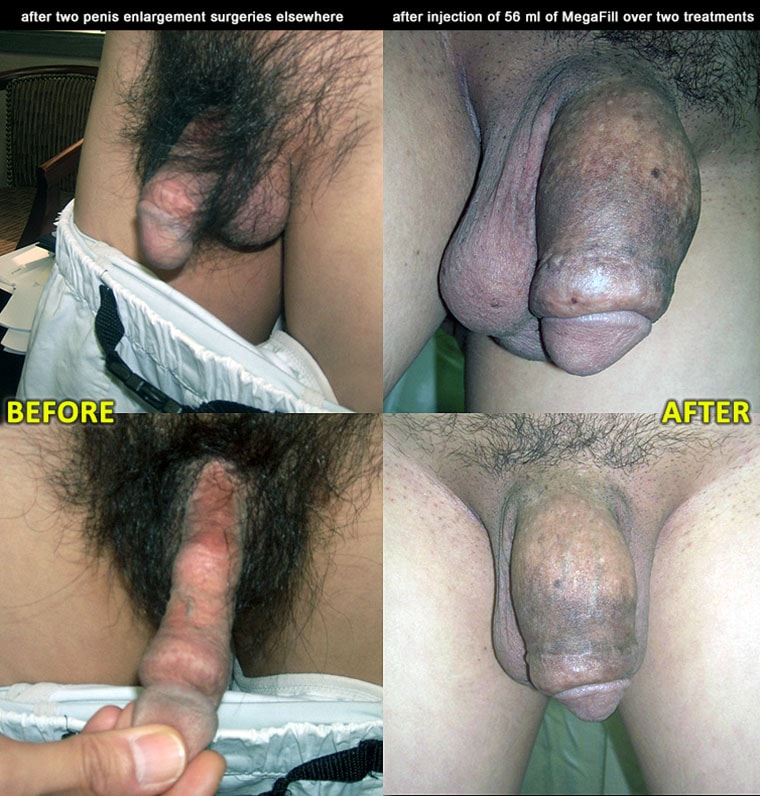
What does the doctor recommend?
Why Dr Kim recommends MegaFill Phalloplasty
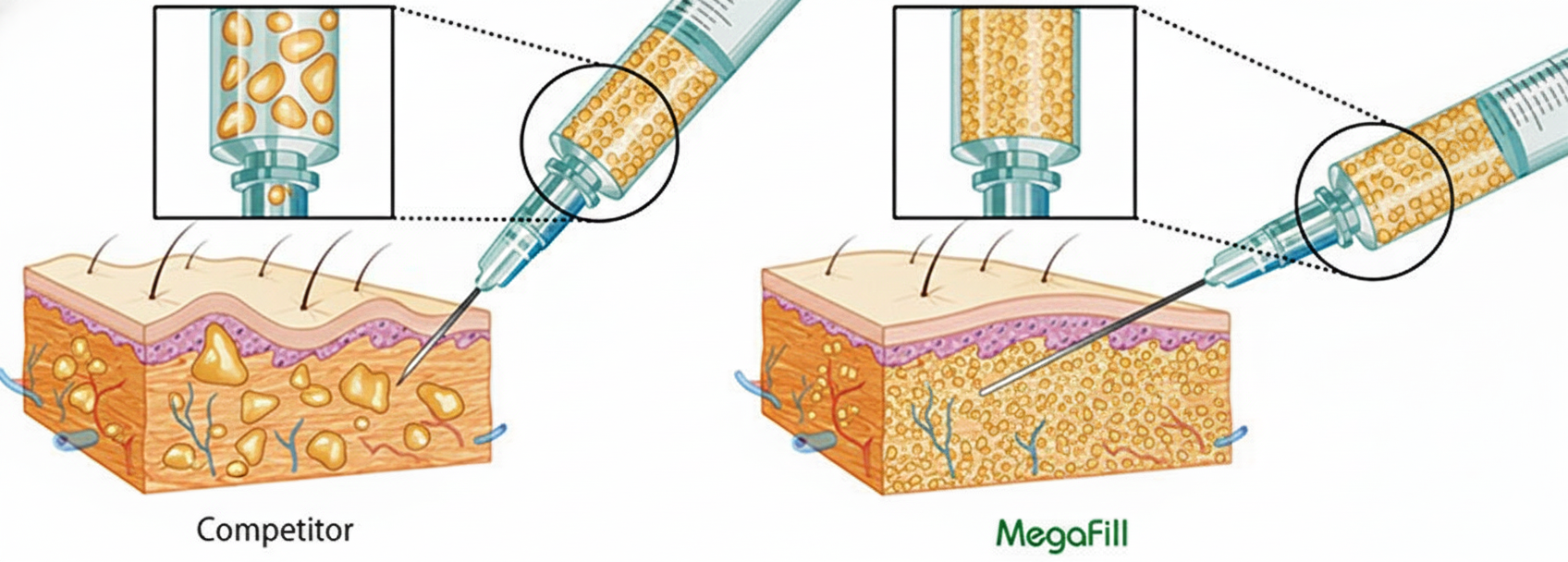
MegaFill is an injectable scaffold derived from donated human tissue and used in phalloplasty to support connective tissue growth. The scaffold and the body's regenerative response combine to increase penile girth in selected patients.
This approach leverages tissue engineering: the biologic scaffold orients cellular ingrowth and collagen deposition to produce a durable augmentation. Clinicians refer to the lasting effect as the product's “final cumulative gain.”